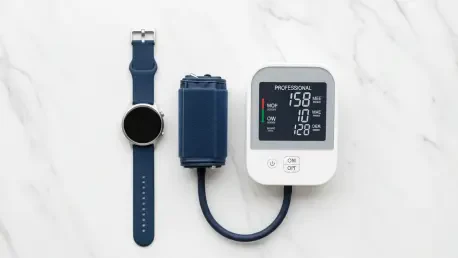
The emergence of sophisticated medical-grade sensors within consumer wearables has fundamentally reshaped how individuals in the United States interact with their cardiovascular health data on a daily basis. For many years, the ability to track blood pressure directly from the wrist was a restricted luxury, available in dozens of countries but legally sidelined in the American market. The recent activation of this feature for the Galaxy Watch ecosystem finally brings a long-awaited diagnostic-adjacent tool to millions of users who require consistent health insights.
This article serves as a detailed examination of the operational mechanics, regulatory history, and technical requirements that define the Samsung Health Monitor experience. Readers can expect to learn exactly which devices are compatible, why a traditional cuff remains a necessary part of the process, and how this data compares to that provided by other major tech competitors. By exploring these facets, the objective is to provide a clear roadmap for navigating the complexities of wearable-based hypertension management.
Introduction
The integration of blood pressure monitoring into the Samsung Galaxy Watch series represents more than just a software update; it is a pivotal response to a widespread domestic health crisis. With a significant portion of the adult population in the United States dealing with hypertension, the demand for accessible, non-invasive monitoring tools has never been higher. The activation of this feature provides a bridge between clinical visits and daily life, allowing for a more granular view of cardiovascular trends that were previously difficult to capture outside of a doctor’s office.
Moreover, the rollout highlights the intricate dance between high-tech innovation and federal safety standards. While the hardware for blood pressure tracking has existed in these watches for several generations, the journey toward regulatory compliance in the U.S. was notably prolonged. This article explores the nuances of this transition, ensuring that users understand both the power and the limitations of the sensors strapped to their wrists.
Key Questions or Key Topics Section
What Are the Technical Prerequisites for Accessing Blood Pressure Data?
Utilizing the blood pressure feature requires a specific synergy between hardware and software that is currently limited to the Samsung ecosystem. The functionality is active for the Galaxy Watch 4 and all subsequent models, including the most recent iterations, provided they are updated to the latest version of the wearable operating system. This technological baseline ensures that the onboard optical sensors have the necessary processing power and sensitivity to detect the subtle pulse wave patterns required for blood pressure estimation.
However, the “walled garden” approach remains a significant factor for potential users. To access the Samsung Health Monitor app, which houses the blood pressure data, the watch must be paired with a Samsung Galaxy smartphone running a modern version of Android. This restriction is primarily due to the specific software integrations and security protocols required by the manufacturer to meet regulatory standards. Consequently, those using the Galaxy Watch with non-Samsung Android devices will find this specific health feature remains inaccessible, regardless of the watch’s hardware capabilities.
How Does the Calibration Process Ensure Measurement Accuracy?
The Galaxy Watch does not function as a standalone replacement for medical equipment but rather as a sophisticated extension of it. To maintain accuracy, the system relies on a hybrid methodology involving a traditional upper-arm blood pressure cuff. Every 28 days, users must perform a calibration ritual where they take simultaneous readings from both the watch and the manual cuff. By entering the cuff’s numerical results into the app, the watch can adjust its algorithms to account for the user’s specific arterial characteristics.
Once this baseline is established, the watch uses Photoplethysmogram (PPG) sensors to analyze the relationship between the calibration value and the change in blood pulse waves. This technique allows the device to estimate systolic and diastolic numbers between calibration cycles. While this provides highly convenient trend analysis, Samsung emphasizes that these readings are intended for personal tracking and should not be used to alter medication or replace professional medical advice. The reliance on monthly calibration serves as a vital safeguard to ensure the sensors do not drift into inaccuracy over time.
Why Did the Feature Take Several Years to Reach American Users?
The timeline for the release of blood pressure monitoring in the United States was famously delayed compared to global markets. While the feature was available in regions like Europe and Canada as early as the beginning of the decade, the U.S. market faced a much more rigorous path toward approval. This discrepancy was largely rooted in the stringent evaluation process of the Food and Drug Administration (FDA), which requires extensive data proving both the safety and the reliability of software as a medical device.
The silence from the manufacturer during this multi-year wait led to significant consumer frustration, as the hardware was essentially dormant in a region with high hypertension rates. The eventual activation of the service suggests that the necessary thresholds for clinical accuracy and data privacy have finally been met. This arrival marks a turning point where the digital health capabilities of American wearables are finally catching up to their international counterparts, providing a necessary tool for the 120 million adults currently navigating cardiovascular challenges.
How Does Samsung’s Approach Differ From Other Major Competitors?
When comparing Samsung’s blood pressure monitoring to the solutions offered by competitors like Apple, a fundamental difference in philosophy becomes apparent. Samsung focuses on providing active, discrete numerical data. By giving the user specific systolic and diastolic values, the Galaxy Watch functions as a digital diary for those who need to report exact numbers to their healthcare providers. This approach favors the proactive user who is comfortable with the maintenance of a monthly calibration schedule.
In contrast, other major competitors have often leaned toward passive monitoring and long-term trend identification. Rather than giving a specific blood pressure reading on demand, these alternative systems might monitor blood vessel patterns over several weeks to alert the user of potential hypertension signs. Samsung’s decision to provide direct numbers gives it a distinct advantage for individuals who already have a diagnosis and require more granular, daily data. However, this utility comes with the caveat of the Samsung-only device requirement, forcing users to commit fully to the brand’s ecosystem to reap the benefits.
Summary or Recap
The activation of blood pressure monitoring in the United States transformed the Galaxy Watch from a standard fitness accessory into a specialized health companion. By leveraging a combination of optical sensor technology and manual cuff calibration, Samsung provides a level of detail that was previously unavailable to American wearable users. This system empowers individuals to take a more active role in their cardiovascular health, provided they possess the compatible Samsung hardware and are willing to adhere to the necessary calibration cycles.
Key insights from this rollout include the importance of the 28-day calibration window and the specific focus on numerical data over passive trends. While the regulatory delay was extensive, the resulting availability of the feature offers a vital resource for a population heavily impacted by hypertension. As the market continues to evolve, the integration of such high-stakes medical data into everyday devices signals a move toward more comprehensive personal health management.
Conclusion or Final Thoughts
The journey of Samsung’s blood pressure monitoring in the U.S. demonstrated the complexities of bringing medical innovations to the consumer wrist. It was clear that the intersection of software, hardware, and federal regulation required a delicate balance to ensure user safety. For those who already owned compatible devices, the update finally unlocked the full potential of sensors that had remained underutilized for years. This shift encouraged a more proactive stance on health, where data became a bridge between personal wellness and professional medical consultation.
Moving forward, individuals should consider how these tools fit into their broader health strategy. While the current system requires manual initiation and frequent calibration, it laid the groundwork for future advancements in passive tracking. Users were encouraged to share their digital logs with physicians to provide a clearer picture of their health between office visits. Ultimately, the successful deployment of this technology served as a reminder that the future of medicine is increasingly centered on the data we collect during our daily routines.